DEXON PLUS
Features:
• Advanced Formulation
• Possess Broad Spectrum of Activity
• Rapid Onset of Action
• User and Environment Friendly
• Protection with Care
• Leaves no Sticky Residues
• Aldehyde & Phenol Free
Biodegradable
• One Step Cleaning & Disinfection
• Very Economical
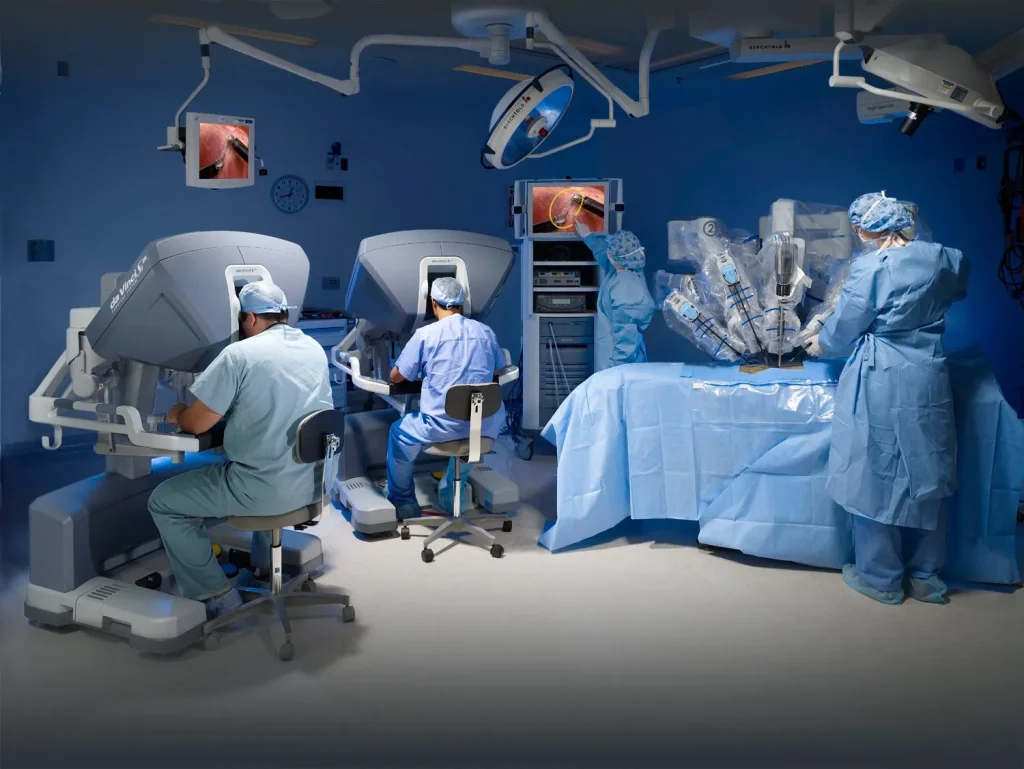
Area of Use:
Ideal for the disinfection between treatments and
therapies, especially where rapid disinfection is
needed. Like Sterile Areas, Quality Control Labs,
Drug Production Areas,Emergency Departments,
Ambulance Services, OPD settings, Minor Surgeries,
Dental Clinics, Endoscopy procedure rooms,
Clinical & Research Labs, Nursing offices, etc.
SPECTRUM OF ACTIVITY Effective against pathogens like:
- Coronavirus (COVID-19)
- Vaccinia Virus (Small Pox)
- HIV (AIDS)
- Hepatitis B Virus (Hep.B)
- Hepatitis C Virus (Hep.C)
- Influenza Virus (Flu)
- Poliovirus (Polio)
- Adenovirus (Fever, Cough, pink eye)
- Simian Virus (Cancer)
- Mycobacterium terrae (TB)
- Mycobacterium avium (TВ)
- Mycobacterium tuberculosis (TB)
- Staphylococcus aureus
- (Skin & Soft tissue Infections)
- Enterococcus hirae (Sepsis)
- Pseudomonas aeruginosa
(Pneumonia) - MRSA (Resistant S. aureus)
- VRE (Resistant Enterococci)
- CRE (Resistant Enterococci)
- Candida albicans (Candidiasis)
- Aspergillus brasiliensis (Aspergillosis)

Matthias la Gordt Dillie
Director | Business Leader
InterChemie - Holland
Interchemie werken ‘De Adelaar’ B.V. was founded on the 19th of January 1979 in Castenray, the Netherlands. From its
humble beginnings, Interchemie is now supplying health products such as medicines, nutritionals, disinfectants, feed
additives, biosecurity and a variety of other products to customers worldwide.
We distribute our products to carefully selected exclusive distributors in more than 135 countries across the globe,
making Interchemie a truly global company.
Our factories in Europe produce multiple medicines in accordance with European GMP and GMP+ standards. They are
frequently inspected by the authorities, resulting in GMP and GMP+ certification.
Interchemie aims to achieve the highest quality standards. We do this through continuous research and development
of new products, by continuously enhancing our product lines and processes, and through development and
investment in our most important asset; our people.
Andrii Gavrylenko
AMEA Regional Manager
InterChemie - Holland
At a time when hygiene, safety, and infection control are more critical than ever, we are proud to introduce our
European-standard disinfectant DEXON PLUS to the Pakistani market. This product is specially formulated to meet the
stringent requirements of the pharmaceutical industry and hospital environments, ensuring superior efficacy and
reliability.
Our goal is to support Pakistan’s healthcare and pharma sectors with a high-quality, CE-certified solution that
guarantees cleanliness and microbial control across surfaces, equipment, and sensitive areas such as operation
theatres, wards, and production zones.
With our extensive industry experience and a deep commitment to public health, we believe this product will play a
pivotal role in elevating disinfection standards and contributing to a safer, healthier Pakistan.
Dr. Najmul Hasnain Shah
Global Technical | Product Marketing Manager
InterChemie - Holland
As the Technical representative of InterChemie, I take great pride in presenting our latest innovation-a high-efficacy,
European-certified disinfectant meticulously formulated to meet the stringent hygiene requirements of the
pharmaceutical and healthcare sectors.
This product has been carefully engineered to ensure superior microbial control while maintaining material
compatibility and operator safety. Its non-corrosive nature and rapid action make it an ideal solution for surface and
equipment cleaning within pharmaceutical manufacturing environments, as well as for use in hospital wards, intensive
care units, and operation theaters.
We have rigorously tested this disinfectant to ensure it delivers consistent results in critical cleanroom settings and
clinical spaces. It complies with international standards and is supported by strong validation data to ensure both
efficacy and safety.
